Verification Workflows Built for Compliance-Heavy Labs
CoreNexis helps teams manage verification steps for incoming reagent lots with visibility into what is pending, what has been completed, and what still requires follow-up before material is cleared for use.
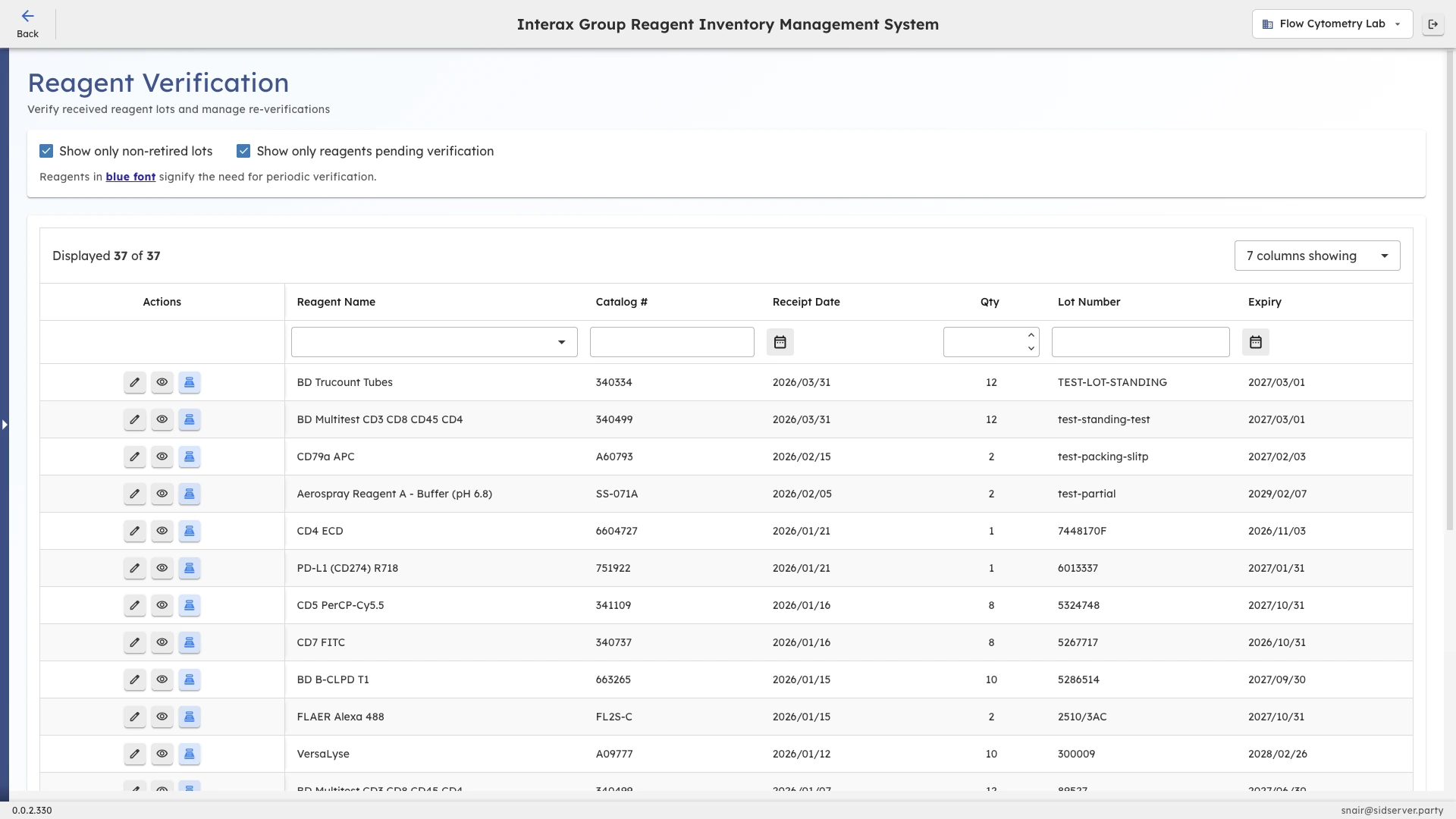
The Challenge
In compliance-heavy laboratories, every incoming reagent lot must pass through verification steps before it can be used on patient samples or in testing workflows. In practice, these steps often live in paper logbooks, shared documents, or the institutional memory of experienced staff. When someone is out sick or a new technician joins, verification gaps appear silently. Lots get used before checks are complete, or completed verifications go unrecorded. The result is audit findings, investigation time, and a nagging uncertainty about whether every lot in active use has actually been cleared.
How Verification Workflow Works
Lot-by-lot verification tracking
Each reagent lot in CoreNexis carries its own verification record covering what steps are required, what has been completed, and what is still outstanding. This is not a shared checklist or a note in a spreadsheet. It is a persistent, lot-specific status that updates as staff work through the process and remains visible to anyone who needs to check the state of a given lot.
Status-driven oversight
CoreNexis surfaces verification status alongside other operational context like receipt date, expiry window, and current stock level so supervisors and quality leads can prioritize what to review first. Instead of pulling reports or checking with individual staff, the information is available in the normal flow of work, reducing the overhead of staying on top of verification queues.
Compliance-ready records
When an audit or internal review requires documentation of verification activity, the records are already structured, timestamped, and organized by lot. There is no assembly step. No pulling data from three systems, no reformatting spreadsheets. The verification history for any lot or time period is already in a form that supports the questions reviewers actually ask.
What Changes
- Keep verification tasks visible instead of buried in ad hoc notes.
- Create a clearer audit trail for each lot under review.
- Reduce the risk of lots moving forward without the right checks completed.
See Verification Workflow in your lab's context
We can walk through the workflows, reporting, and operational controls most relevant to your environment.
