Reporting That Supports Expiry Control and Compliance Readiness
CoreNexis helps laboratories generate reporting views that support expiry management, review readiness, stock oversight, and operational follow-through across compliance-sensitive inventory processes.
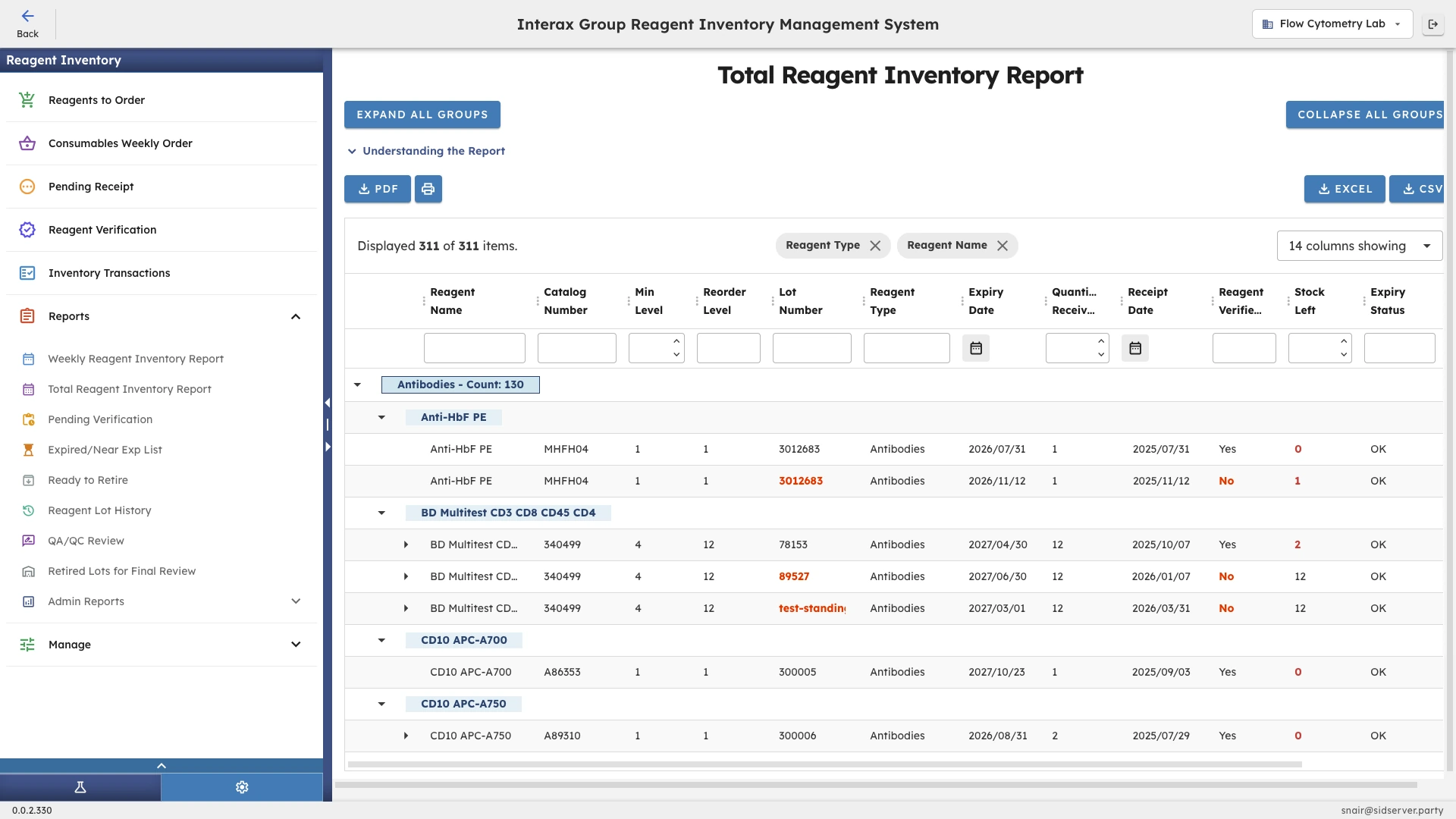
The Challenge
Managing reagent expiry is one of those tasks that seems straightforward until it is not. Labs deal with hundreds or thousands of lot entries, each with its own expiry date, verification state, and usage status. Keeping track of what is approaching expiry, what has already expired, and what needs to be pulled from active use requires constant attention, and when that attention lapses, the consequences range from wasted inventory to compliance findings. Most teams manage this with filtered spreadsheets, calendar reminders, or periodic manual reviews, all of which degrade as inventory complexity grows.
How Compliance Reports Works
Inventory-wide reporting
CoreNexis generates reporting views that cover the full breadth of the laboratory's reagent inventory including stock levels, expiry dates, lot detail, verification status, and usage history in a single, filterable output. These are not canned reports with fixed fields. Teams can adjust what they see based on the question they are trying to answer, whether that is what expires in the next 30 days or which lots in a given category are still pending verification.
Compliance-oriented views
The reporting structure in CoreNexis is built around the questions that regulatory bodies, quality teams, and internal reviewers actually ask. Rather than exporting raw data and reshaping it for each audience, the system organizes output by the dimensions that matter for compliance conversations: lot traceability, verification completeness, expiry status, and operational follow-through.
Operational follow-up
Reports in CoreNexis are not endpoints. They are starting points for action. When a report surfaces a near-expiry lot, an incomplete verification, or a gap in receiving records, the information is presented with enough context to act on it directly. The goal is to make reporting a tool for operational improvement, not just a compliance checkbox.
What Changes
- Reduce last-minute scrambling around expiry and reporting deadlines.
- Create clearer reporting outputs for internal review and external oversight.
- Give teams a more reliable picture of stock condition and verification state.
See Compliance Reports in your lab's context
We can walk through the workflows, reporting, and operational controls most relevant to your environment.
